DAYVIGO is a dual orexin receptor antagonist with convenient dosing, once nightly right before bed, with at least 7 hours remaining before planned awakening.

| Dose Adjustment | Dose Adjustment | |||
|---|---|---|---|---|
| No Adjustment | Use 5 Mg Only | Not Recommended | ||
| Pediatrics | ||||
| Geriatrics | ||||
| Hepatic Impairment | Mild | |||
| Moderate | ||||
| Severe | ||||
| Renal Impairment | ||||
| CYP3A Inhibitors | Weak | |||
| Moderate | ||||
| Strong | ||||
| CYP3A Inducers | Weak | |||
| Moderate | ||||
| Strong | ||||


Recommend an appropriate trial period
Counsel your patients on good sleep hygiene
In addition to treatment, patients may benefit from improving their sleep hygiene—habits that can help them optimize sleep.
Some examples can include:
Learn about a study that evaluated transition from a commonly prescribed insomnia medication to DAYVIGO
An open-label pilot study to assess the dosing approach of directly transitioning from zolpidem (Zol) to DAYVIGO 5 mg or 10 mg
Basic Demographics
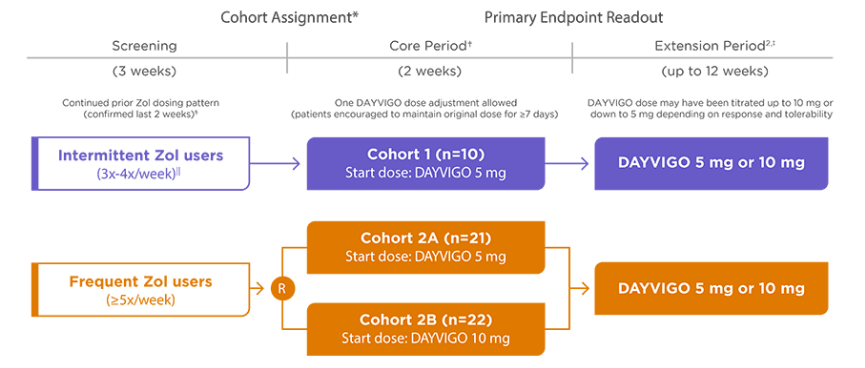
ER=extended release; IR=immediate release; R=randomization; Zol=zolpidem tartrate.
⁎ Patients did not taper their study entry Zolpidem dose.
† Follow-up visit occurred 4 weeks after completion of the core period (or as soon as possible following early discontinuation) for patients who did not enter the extension period.
‡ Follow-up visit occurred 4 weeks after last dose of DAYVIGO for those who completed or discontinued during the extension period.
§ History (≥1 month) of intermittent (3-4 times/week) or frequent (≥5 times/week) use of Zol-IR (max 10 mg/night) or Zol-ER (max 12.5 mg/night), and not taking a dose that was lower than what was prescribed.
∥ Patients who met both criteria for intermittent and frequent Zol use for 1 week each of the last 2 weeks of the 3-week screening period were assigned to cohort 1 and referred to as cohort 1-mixed.
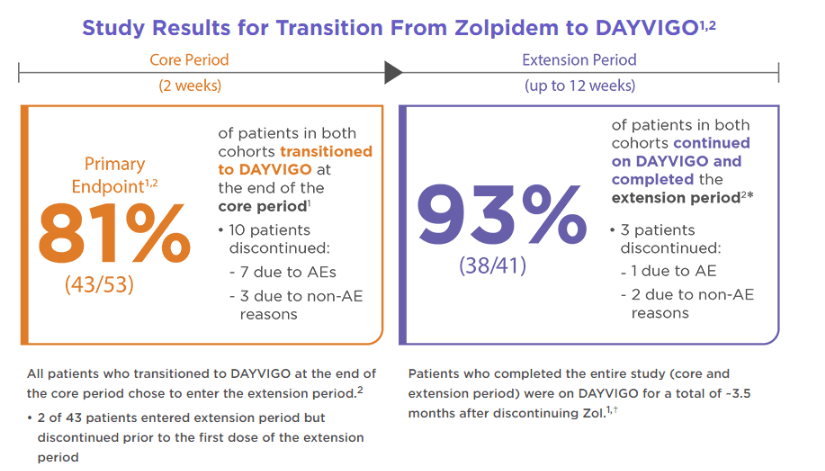
AE=adverse event; Zol=zolpidem tartrate.
⁎Of the patients who entered and received treatment during extension period, 9 were in cohort 1 and 32 were in cohort 2.
†Patients were allowed to select the days they wanted to take a dose.
| Safety Assesments | Core Period | Extension Period | ||
|---|---|---|---|---|
| DAYVIGO5 mg*( n=36) | DAYVIGO10 mg*( n=37) | DAYVIGO5 mg*( n=32) | DAYVIGO10 mg*( n=37) | |
| Treatment-related TEAEs, n | 2 | 10 | 0 | 9 |
| TEAE leading to study drug discontinuation | 2 | 5 | 0 | 1 |
| TEAEs with incidence >5% in either cohort 1 or 2, n | ||||
| Abnormal Dreams | 0 | 4 | 0 | 2 |
| Somnolence | 1 | 3 | 0 | 3 |
| Conjunctival hemorrhage | 0 | 1 | 0 | 1 |
| Sinusitis | - | - | 0 | 1 |
| ECG - QT prolongation | 1 | 0 | 1 | 0 |
| Accidental overdose | 2 | 0 | - | - |
| Intentional overdose | 1 | 1 | - | - |
| Product admin error | 0 | 1 | 1 | 0 |
AE=adverse event; COVID-19=coronavirus disease 2019; ECG=electrocardiogram; SAE=serious adverse event; TEAE=treatment-emergent adverse event; Zol=zolpidem tartrate.
⁎On-treatment dose, ie, dose taken at the time of AE.
†TEAEs considered by the investigator to be related to study drug or TEAEs with missing causality.
‡Both patients were recorded as having intentional overdose to improve sleep and were declared to be mild in severity and nonserious.
§Two patients experienced more than 1 AE.